Not All Plastics Are Created Equal: What to Avoid?
Author: Jian Gao, PhD
Editor: Mr. Frederick Malphurs
August 31, 2025
The Big Picture
In recent years, microplastics in the environment have garnered significant media coverage and public attention. However, they are only the tip of the iceberg – the toxic chemicals leaching from plastics in consumer products pose an even greater danger to our health. In fact, we have been living with a giant double-edged sword: While plastics are ubiquitous, useful, and in some cases indispensable, they constantly leach toxic chemicals into the air we breathe, the water we drink, and the food we eat – poisoning us in the process.
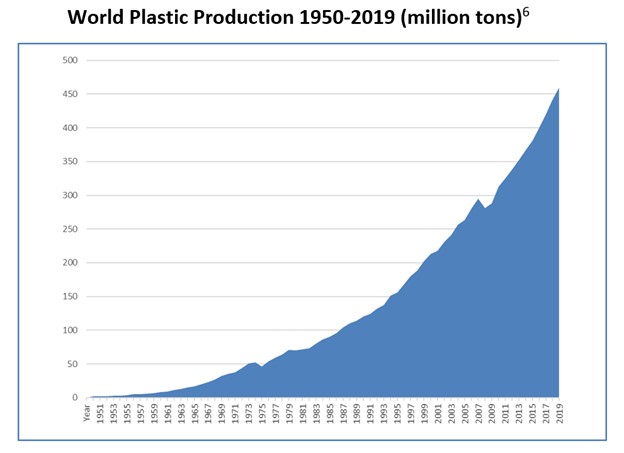
Since World War II, plastic production has increased exponentially. Global plastic production has surged from 2 million tons in 1950 to over 450 million tons today—an astounding 225-fold increase.1
Avoiding plastics today is extremely challenging. Without them, how would we brush our teeth, turn on the TV, make a phone call, text our friends, use a tablet or computer, or drive a car? If all plastics were removed from our homes, we would be forced to return to the Stone Age. Plastics undoubtedly offer immense convenience, but their overuse, especially in the food industry, has become a curse to the environment, wildlife, and human health.
Obviously, we cannot avoid all plastics. To steer clear of the most harmful ones, we first need to understand what they are.
The word “plastic” is derived from the Greek word plastikos, meaning “fit for being shaped or molded.” Plastics are polymers—long chains composed of many repeated molecular units. These molecular units are typically hydrocarbons, made up of hydrogen and carbon. The raw materials for most synthetic plastics are derived from crude oil, natural gas, or coal.
The properties of plastics depend in part on the structure and length of their polymer chains. For instance, loosely spaced polymers create softer or more pliable plastics, while densely packed ones result in rigid plastics. At first glance, plastics don’t seem too bad—they largely consist of seemingly harmless hydrogen and carbon atoms. However, the issue lies in the fact that polymers alone rarely meet the physical requirements for practical use. To achieve desirable properties such as color, flexibility, or strength, a variety of chemicals are added during production. Unfortunately, these additives often pose greater health risks than the plastics themselves.
The world saw its first true synthetic plastic, Bakelite, in 1907, thanks to Leo Hendrik Baekeland, who is regarded as the father of plastics. Bakelite is made from formaldehyde and phenol, both of which are highly toxic.
Formaldehyde, which has been extensively studied, is a cancer maker. In 2011, the National Toxicology Program of the Department of Health and Human Services classified formaldehyde as a known human carcinogen in its 12th Report on Carcinogens.2
Formaldehyde is a colorless, strong-smelling, and flammable chemical. In addition to its use in plastics, formaldehyde is commonly found in building materials and many household products, including pressed-wood items (e.g., particleboard, plywood, and fiberboard), glues and adhesives, permanent-press or wrinkle-resistant fabrics, paper product coatings, some insulation materials, fungicides, germicides, disinfectants, and preservatives used in medical labs. Its prevalence makes it nearly ubiquitous.
Phenol is a toxic, colorless crystalline solid with a sweet, tarry odor reminiscent of a hospital smell. It can harm the central nervous system and heart, potentially causing dysrhythmia, seizures, and even coma.3 Long-term exposure may damage the liver and kidneys.4 Yet, phenol remains a key active ingredient in sore throat sprays. Fortunately, there is currently little or no evidence linking it to coronary heart disease or cancer.5
Beginning in the early 1960s, Bakelite was rapidly replaced by newer plastics with more desirable properties, such as bright colors and increased strength. Simultaneously, the production and use of plastics skyrocketed as shown in the figure below. The American Chemical Society proudly proclaimed: “Today, synthetic plastics are everywhere. They are as familiar to us as wood or metal and are easily taken for granted. Almost anyone can name a dozen familiar products made in part or in whole with plastic: toys, computers, clothing, sports equipment, carpet, appliances, building materials, signs, office supplies, packaging, phones, and fashion accessories.”
Plastics by Resin Identification Code (RIC)
Understanding the dangers of plastics can be overwhelming due to their sheer variety and prevalence. One of the simplest ways to assess the toxicity of plastics is by referring to the Resin Identification Code (RIC), also known as the recycling code. This code is represented by a triangle enclosing a number (1 through 7), often found on the bottom of plastic products, such as bottles.
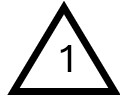 Polyethylene terephthalate (PET)
Polyethylene terephthalate (PET)
Probably this is the most well-known plastic, which initially gained fame as a wrinkle-free fiber (commonly called polyester). It is widely used for food and beverage packaging—such as water, soft drinks, juice, salad dressing, medication bottles, and peanut butter or jam jars—as well as microwaveable and oven-ready meal trays. Additionally, PET is used to make containers for household products like detergent and cleaner bottles, and to fill pillows and comforters. It is everywhere.
PET has been shown to leach antimony, a toxic metal used as a catalyst and flame retardant. Antimony is associated with several health problems, including lung and cardiovascular diseases, as well as liver and reproductive disorders.7-10 Antimony is classified as possibly carcinogenic to humans (Group 2B) by the International Agency for Research on Cancer.11
We are all exposed to low levels of antimony in the environment, as it is a naturally occurring metal typically found in ore deposits. As a result, it can be present in soil, water, and air. However, additional exposure from water bottles and food containers could jeopardize our health. Research has clearly shown that the longer water or food stays in these containers, the more leaching of chemicals occurs, and the level of leaching increases exponentially as the temperature rises.12-14
Research also indicates phthalates leach from PET.13-15 Phthalates, often referred to as plasticizers, are a group of chemicals used to make plastics more flexible and durable. These chemicals are also found in hundreds of products, such as vinyl flooring, adhesives, detergents, and personal-care items (e.g., soaps, shampoos, hair sprays, and nail polishes). Over the last decade, researchers have linked phthalates to a variety of health issues, including autism spectrum disorders, attention deficit hyperactivity disorder (ADHD), low IQ, other neurodevelopmental problems, asthma, obesity, type II diabetes, altered reproductive development, breast cancer, and cardiovascular diseases.16-20 Phthalates are among the “dirty dozen” endocrine disruptors.21
In fact, the worst part is the unknown—nobody knows exactly what is inside plastics or what’s leaching out, as manufacturers aren’t required to report the additives used in PET or other plastics. But one thing we do know for sure: “Almost all commercially available plastic products we sampled—independent of the type of resin, product, or retail source—leached chemicals with reliably detectable estrogenic activity (EA), including those advertised as BPA-free. In some cases, BPA-free products released chemicals with more EA than BPA-containing products,” concluded a study published in the well-respected Journal of Environmental Health Perspectives.22
Estrogenic activity refers to chemicals or additives used in plastics that imitate the sex hormone estrogen in our body, tricking the body into thinking they are the real thing. The consequences are serious: it causes a range of health problems, “such as early puberty in females, reduced sperm counts, altered functions of reproductive organs, obesity, altered sex-specific behaviors, and increased rates of some breast, ovarian, testicular, and prostate cancers.”23
Take, for example, a study conducted at the University of Missouri, which showed that a bottled water brand triggered a 78% increase in the growth of breast cancer cells compared to the control sample. The frustrating part is that researchers can’t pinpoint which chemicals caused the problem. However, they do know for sure that the cancer-promoting chemicals mimic estrogen, a hormone linked to breast cancer, because when estrogen-blocking chemicals were added, the cancer cell proliferation was inhibited.24
The tricky part is that not all PET plastics are the same. Lab tests have shown that the estrogenic activity from PET bottles varies widely from brand to brand.25,26 What we get from PET largely depends on what manufacturers put into their products in the first place.
To avoid the harmful chemicals in PET, the best policy is to avoid all food and drink in plastic containers. If that’s not possible, at least avoid buying water and food in plastic containers and letting them sit for too long, and don’t leave them in hot places like garages or cars.
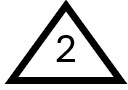 High-density Polyethylene (HDPE)
High-density Polyethylene (HDPE)
HDPE is one of the most widely used families of plastics in the world. It has the simplest basic chemical structure of any plastic polymer—long, virtually unbranched polymer chains with repeating units of CH₂ (one carbon and two hydrogen atoms), which makes it very strong and easy to process. It is widely used to make water, milk, and juice containers, cereal box liners, yogurt and margarine tubs, bleach, detergent, and shampoo bottles, and garbage bags, to name a few. HDPE is generally considered safe, or safer than other plastics such as PET. However, studies have also shown that nonylphenol (NP), an endocrine disruptor, and other estrogen-mimicking chemicals can leach from HDPE products.22.27.28
Again, the scariest part is the unknown—these additives. The chemical composition of almost all commercially available plastics is proprietary and unknown to the public. A baby bottle, for example, can consist of over 100 chemicals, most of which can leach from the product.22
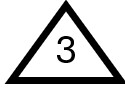 Polyvinyl Chloride, Commonly Known as PVC or Vinyl
Polyvinyl Chloride, Commonly Known as PVC or Vinyl
PVC is another widely produced and used plastic. PVC can be made rigid or flexible, depending on the additives used. PVC pipes are best known for their rigid form, while toys and clothes are best known for their soft form. PVC remains popular because of its versatility and low production cost. However, its use, especially in developed countries, has decreased due to serious health and environmental risks.
PVC is probably the most toxic plastic still used to make consumer goods. The toxicity of vinyl chloride, from which PVC is made, was recognized as early as the 1940s.29-31 As summarized by the EPA, vinyl chloride causes serious damage to every major organ or system in the human body.32 The EPA has classified vinyl chloride as a Group A human carcinogen—it causes liver cancer.33 Just when you think it couldn’t get worse, it does: the additives in PVC are even more unpredictable. Beyond the unknowns, among the known additives, two phthalates (DEHP and BBzP) commonly used in manufacturing soft forms of PVC for toys and bottles are endocrine disruptors mimicking estrogen. These phthalates have also been linked to allergic disorders, asthma, ADHD in children,34-36 and breast cancer in women.37
The good news is that the use of these dangerous phthalates in children’s products is restricted. The bad news is they are still used elsewhere – from the tubs used in hospitals to some food packaging. “Banned chemicals still used in hospital IVs linked to attention deficit disorder,” reported the Washington Post. “Phthalates are out of infants’ toys, but a heavy dose is still in their food.”38-40
The bottom line: Try your best to stay away from PVC.
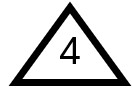 Low-density Polyethylene (LDPE)
Low-density Polyethylene (LDPE)
LDPE and HDPE are cousins – both are types of polyethylene (PE), the most widely used plastic in consumer products. Globally, about 80 million tons of PE are produced every year. The major difference between LDPE and HDPE, as indicated by their names, lies in their density, which results from the structure of their polymer chains. The chains in HDPE are unbranched, while in LDPE, they are branched. The branched chains in LDPE make it less dense and more flexible.
LDPE is widely used for grocery and other household bags, food storage containers (such as squeezable bottles for honey or mustard), and as a coating for paper milk cartons and hot & cold beverage cups. LDPE shares a similar safety and health-risk profile with HDPE.
 Polypropylene (PP), Also Known as Polypropene
Polypropylene (PP), Also Known as Polypropene
PP is a plastic with a simple chemical structure consisting of many methyl groups (CH₃ — one carbon and three hydrogen atoms) linked together. Like HDPE, PP is very stable at room temperature – resistant to fats and almost all organic solvents – which makes it suitable for food containers. It is also widely used for automobile parts, such as bumpers.
PP is generally considered a safer plastic for food and drink use. However, “safer” does not mean completely safe. Few studies have investigated the health effects of PP under normal daily use. A study published in Science demonstrated that disposable PP labware, such as test tubes and pipette tips, leaches plastic additives like oleamide, a plastic stabilizer. In one case study, the breakdown products of PP were found to trigger asthma. Additionally, researchers have shown that most plastics, including PP, release estrogenic chemicals when tested with solvents.41
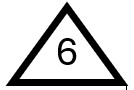 Polystyrene (PS)
Polystyrene (PS)
PS can be foamed or solid, depending on the additives and manufacturing process. In its solid form, it is often used to make labware such as test tubes and petri dishes, as well as everyday items like CD cases and smoke detector housings. Foamed PS is ubiquitous: when opening boxes containing fragile items such as TVs or tablets, you often see white foam used as cushioning. It is also widely used as insulation for appliances and buildings. PS gets uncomfortably close to our food, as it is commonly used to make egg cartons, disposable bowls, cups, deli food plates, and take-out food containers.
Don’t be fooled by its benign appearance – PS is highly toxic. PS food containers can leach styrene (a derivative of benzene), which is associated with serious health issues, including brain damage and cancer.42-44 The EPA describes styrene as “a suspected toxin to the gastrointestinal tract, kidney, and respiratory system, among others,” and the U.S. National Toxicology Program has classified it as “reasonably anticipated to be a human carcinogen.”45,46
The takeaway is clear: stay away from food served in these puffy plastic bowls, plates, cups, or boxes.
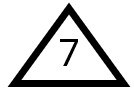 Other
Other
If a plastic does not fall into any of the six primary categories, it is classified as type 7. Despite 7 being considered a lucky number, many manufacturers now avoid labeling their products with it due to the negative publicity surrounding BPA (Bisphenol A), which falls under this category. However, not all plastics labeled as 7 are harmful. A newer type of plastic made from bio-based polymers, such as corn starch, is compostable and also classified as 7, often labeled with “PLA” or “Compostable” alongside the number. Unfortunately, these eco-friendly plastics are not yet mainstream because they are more expensive to produce compared to petroleum-based plastics.
In recent years, BPA has become one of the most infamous plastics. BPA, which is also used as an additive in other plastics, is the primary component in polycarbonate (PC) and epoxy, both of which are classified as type 7 and typically labeled with “PC” beneath the recycling symbol. Polycarbonate was widely used to make water bottles and baby products, while epoxy was commonly used to coat the interiors of metal products, such as food cans, bottle caps, and water supply lines. However, its use in consumer products has drastically decreased in recent years due to concerns about BPA’s potential adverse health effects.
Mounting evidence shows BPA is an endocrine disruptor, mimicking human hormones such as estrogen, which play crucial roles in cellular functions. Interestingly, BPA, first synthesized in 1891 by a Russian chemist, was initially used as synthetic estrogen in the 1930s. Later, chemists discovered that when mixed with phosgene (a toxic gas used during World War I) and other chemicals, BPA transformed into a clear, shatter-resistant plastic.47 BPA-based plastics have been in commercial use since 1957, with few questioning their safety for human health until 1998, when their harmful effects were discovered by accident.
Patricia Hunt, a geneticist at Washington State University, was dubbed “the accidental toxicologist” by Scientific American. For over 25 years, her research focused on how age affects women’s ability to produce genetically normal eggs. In 1998, while at Case Western Reserve University in Cleveland, Ohio, Hunt stumbled upon a finding that not only altered her career but also shifted public perception of plastics. While preparing to publish her findings on female mice’s hormone levels and egg abnormalities, Hunt needed to confirm that the eggs of the control group were normal. To her surprise and dismay, she discovered that 40% of the mice in the control group had egg defects.
Instead of publishing her paper, Hunt spent the next four months investigating the cause of the problem. The culprit turned out to be BPA. A temporary worker in the animal facility had used the wrong detergent to wash cages and water containers, which caused BPA to leach out and expose the mice to it. This unforeseen incident shifted Hunt’s focus toward the study of plastic toxicity. In subsequent studies, she and her colleagues found that “low-dose BPA exposure during pregnancy has multigenerational consequences; it increases the likelihood of chromosomally abnormal grandchildren.”48
This discovery marked the beginning of widespread attention to BPA from both researchers and the public. Since then, nearly fourteen thousand papers on BPA have been published. The majority of studies have demonstrated that BPA is linked to a myriad of health problems, including reproductive and neurological disorders, chromosome damage in female ovaries, decreased sperm production in males, early onset of puberty, cardiovascular and metabolic diseases, obesity, type 2 diabetes, and various cancers.49,50
However, controversies over the findings persist. Generally, studies funded by the plastic industry conclude that low-dose BPA is safe for humans, whereas independent, non-industry-funded studies often find otherwise. For instance, a review of animal and human studies on low-dose BPA exposure, published in Environmental Health Perspectives, revealed that 90% (94 out of 104) of non-industry-funded studies found low-dose BPA to be harmful, while 100% (11 out of 11) of industry-funded studies found it to be harmless.51
A significant challenge in studying BPA’s health effects lies in the limitations of traditional toxicological analysis. Conventional toxicology operates on the principle that a causal relationship between exposure and outcome requires a dose-response pattern – meaning the higher the exposure, the worse the outcome. The issue with BPA is that it does not conform to this rule. Low-dose exposure often induces adverse effects on the endocrine system, cardiovascular system, and tumor growth, while high-dose exposure can suppress the body’s response and sometimes produce entirely different effects.52-54
Thus far, the FDA’s official position is that low-dose exposure to BPA is safe for human health. Consequently, there is no federal law banning the use of BPA in any products. However, several states, including Connecticut, Maryland, Minnesota, Vermont, Washington, and Wisconsin, have enacted restrictions on the sale of certain products, such as bottles and sippy cups containing BPA. To mitigate negative publicity, many manufacturers have voluntarily stopped using BPA in baby products.
The good news for consumers is that while researchers and regulators continue testing and debating, manufacturers have largely phased out BPA in food containers and water bottles –leading to the prevalence of “BPA Free” labels. The bad news, however, is that the safety of the chemicals used to replace BPA remains unknown.
What to Avoid?
Regardless of whether a product is labeled “BPA free” or not, the bottom line is that no plastic is entirely safe. From an evolutionary perspective, the human body is not equipped to process the chemicals in plastics, which have only been part of our environment for the past 100 years. Taking a “better safe than sorry” approach could be a lifesaver – it’s too late to act once a serious health problem arises. Whenever possible, avoid food stored in plastic containers and minimize the consumption of drinks from plastic bottles.
If you must buy or use plastic products, those with recycling codes 2, 4, and 5 are considered relatively safer compared to 1, 3, 6, and 7 (PC).
About the Author and Editor:
Jian Gao, PhD – A dedicated healthcare analyst/researcher with 28 years of experience, committed to pursuing truth and evidence over trends and fads.
Frederick Malphurs – A visionary healthcare executive who led multiple hospitals over a distinguished 37-year career, dedicated to advancing patient care and strengthening health systems.
References:
- Ritchie H, Samborska V, Roser M. Plastic Pollution. 2024. https://ourworldindata.org/plastic-pollution#:~:text=Plastic%20production%20has%20sharply%20increased.
- National Toxicology Program (June 2011). Report on Carcinogens, Twelfth Edition. Department of Health and Human Services, Public Health Service, National Toxicology Program. Retrieved June 10, 2011, from: http://ntp.niehs.nih.gov/go/roc12.
- Warner, MA; Harper, JV. Cardiac dysrhythmias associated with chemical peeling with phenol”. Anesthesiology. 1985;62 (3): 366–7.
- World Health Organization/International Labour Organization: International Chemical Safety Cards, http://www.inchem.org/documents/icsc/icsc/eics0070.htm
- Babich H, Davis DL. Phenol: a review of environmental and health risks. Regul Toxicol Pharmacol. 1981 Jun;1(1):90-109.
- https://ourworldindata.org/grapher/global-plastics-production
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Antimony. U.S. Public Health Service, U.S. Department of Health and Human Services, Altanta, GA. 1992.
- S. Department of Health and Human Services. Hazardous Substances Data Bank (HSDB, online database). National Toxicology Information Program, National Library of Medicine, Bethesda, MD. 1993.
- https://www.epa.gov/sites/production/files/2016-09/documents/antimony-compounds.pdf
- Sundar S, Chakravarty J. Antimony Toxicity. Int J Environ Res Public Health. 2010 Dec; 7(12): 4267–4277.
- Summaries & Evaluations Antimony Trioxide and Antimony Trisulfide. International Agency for Research on Cancer; Lyon, France: 1989. [(accessed on 29 January 2010)]. Available online: http://www.inchem.org/documents/iarc/vol47/47-11.html.
- Yang CZ, Yaniger SI, Jordan VC, et al. Most Plastic Products Release Estrogenic Chemicals: A Potential Health Problem That Can Be Solved. Environ Health Perspect. 2011 Jul 1; 119(7): 989-996.
- https://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors#.WyRfSeS0Whc
- Naidenko O, Leiba N, Sharp R, Houlihan J. Bottled Water Quality Investigation: 10 Major Brands, 38 Pollutants. Available: http://courses.washington.edu/h2owaste/assignments/ExtraBottled%20Water.pdf.
- Pinto B, Reali D. Screening of estrogen-like activity of mineral water stored in PET bottles. Int J Hyg Environ Health. 2009;212(2):228–232.
- Jeddi MZ, Janani L, Memari AH, et al. The role of phthalate esters in autism development: A systematic review. Environ Res. 2016 Nov;151:493-504.
- Benjamin S, Masai E, Kamimura N, et al. Phthalates impact human health: Epidemiological evidences and plausible mechanism of action. J Hazard Mater. 2017 Oct 15;340:360-383. Epub 2017 Jun 19.
- López-Carrillo L, Hernández-Ramírez RU, Calafat AM, et al. Exposure to Phthalates and Breast Cancer Risk in Northern Mexico. Environ Health Perspect. 2010 Apr; 118(4): 539–544.
- Mariana M, Feiteiro J, Verde I, Cairrao E. The effects of phthalates in the cardiovascular and reproductive systems: A review. Environ Int. 2016 Sep;94:758-776.
- Jaimes R 3rd, Swiercz A, Sherman M, et al. Plastics and cardiovascular health: phthalates may disrupt heart rate variability and cardiovascular reactivity. Am J Physiol Heart Circ Physiol. 2017 Nov 1;313(5):H1044-H1053.
- https://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors#.W5QtmeS0VPY
- Yang CZ, Yaniger SI, Jordan VC, et al. Most Plastic Products Release Estrogenic Chemicals: A Potential Health Problem That Can Be Solved. Environ Health Perspect. 2011 Jul 1; 119(7): 989-996.
- https://www.ewg.org/research/dirty-dozen-list-endocrine-disruptors#.WyRfSeS0Whc
- Naidenko O, Leiba N, Sharp R, Houlihan J. Bottled Water Quality Investigation: 10 Major Brands, 38 Pollutants. Available: http://courses.washington.edu/h2owaste/assignments/ExtraBottled%20Water.pdf.
- Pinto B, Reali D. Screening of estrogen-like activity of mineral water stored in PET bottles. Int J Hyg Environ Health. 2009;212(2):228–232.
- Wagner M, Oehlmann J. Endocrine disruptors in bottled mineral water: total estrogenic burden and migration from plastic bottles. Environ Sci Pollut Res Int. 2009;16(3):278–286.
- Loyo-Rosales JE, Rosales-Rivera GC, Lynch AM, Rice CP, Torrents A. Migration of nonylphenol from plastic containers to water and a milk surrogate. J Agric Food Chem. 2004 Apr 7;52(7):2016-20.
- Guart A, Bono-Blay F, Borrell A, Lacorte S. Migration of plasticizers phthalates, bisphenol A and alkylphenols from plastic containers and evaluation of risk. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2011 May;28(5):676-85.
- Oster RH, et al. Anesthesia; narcosis with vinyl chloride. Anesthesiology. 1947;8(4):359-361.
- Tribukh SR, et al., Working conditions and measures for their sanitation in the production and utilization of vinyl chloride plastics. Gigiena Sanit. 1949;10:38-45.
- Wagoner JK. Toxicity of vinyl chloride and poly(vinyl chloride): a critical review. Environ Health Perspect. 1983 Oct; 52: 61–66.
- https://www.epa.gov/sites/production/files/2016-09/documents/vinyl-chloride.pdf
- S. Environmental Protection Agency. Health Effects Assessment Summary Tables. FY1997 Update. Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Office of Research and Development, Cincinnati, OH. 1997.
- Bornehag CG, Sundell J, Weschler CJ, et al. The association between asthma and allergic symptoms in children and phthalates in house dust: a nested case-control study. Environ Health Perspect. 2004 Oct;112(14):1393-7.
- Bornehag CG, Lundgren B, Weschler CJ, et al. Phthalates in Indoor Dust and Their Association with Building Characteristics. Environ Health Perspect. 2005 Oct; 113(10): 1399–1404.
- Kim BN, Cho SC, Kim Y, et al. Phthalates exposure and attention-deficit/hyperactivity disorder in school-age children. Biol Psychiatry. 2009 Nov 15;66(10):958-63.
- López-Carrillo L1, Hernández-Ramírez RU, Calafat AM, et al. Exposure to phthalates and breast cancer risk in northern Mexico. Environ Health Perspect. 2010 Apr;118(4):539-44.
- https://www.washingtonpost.com/news/to-your-health/wp/2014/07/08/phthalates-are-out-of-infants-toys-but-a-heavy-dose-is-still-in-their-food/?utm_term=.930c67878876
- http://www.chicagotribune.com/lifestyles/health/ct-banned-chemical-attention-deficit-disorder-20160406-story.html
- Serrano SE, Braun J, Trasande L, et al. Phthalates and diet: a review of the food monitoring and epidemiology data. Environ Health. 2014 Jun 2;13(1):43.
- Westerhoff P, Prapaipong P, Shock E, Hillaireau A. Antimony leaching from polyethylene terephthalate (PET) plastic used for bottled drinking water. Water Res. 2008 Feb;42(3):551-6.
- Ahmad M, Bajahlan AS. Leaching of styrene and other aromatic compounds in drinking water from PS bottles. Journal of Environmental Sciences. 2007;Vol19 (4):421-426.
- Rosengren LE, Haglid KG. Long term neurotoxicity of styrene. A quantitative study of glial fibrillary acidic protein (GFA) and S-100. Br J Ind Med. 1989 May; 46(5): 316–320.
- Huff J, Infante PF. Styrene exposure and risk of cancer. Mutagenesis. 2011 Sep; 26(5): 583–584.
- 12th Report on Carcinogens. https://web.archive.org/web/20110612024850/http://ntp.niehs.nih.gov/index.cfm?objectid=72016262-BDB7-CEBA-FA60E922B18C2540
- https://www.atsdr.cdc.gov/toxfaqs/tf.asp?id=420&tid=74
- Just How Harmful Are Bisphenol A Plastics? Scientific America. Sep 1, 2008. https://www.scientificamerican.com/article/just-how-harmful-are-bisphenol-a-plastics/
- Susiarjo M, Hassold TJ, Freeman E, Hunt PA et al. Bisphenol A Exposure In Utero Disrupts Early Oogenesis in the Mouse. PLoS Genet. 2007 Jan; 3(1): e5.
- Manzoor MF, Tariq T, Fatima B, et al. An insight into bisphenol A, food exposure and its adverse effects on health: A review. Front Nutr. 2022 Nov 3;9:1047827. doi: 10.3389/fnut.2022.1047827. PMID: 36407508; PMCID: PMC9671506.
- NTP Research Report on the Consortium Linking Academic and Regulatory Insights on Bisphenol A Toxicity (CLARITY-BPA): A Compendium of Published Findings. October 2021. https://ntp.niehs.nih.gov/sites/default/files/ntp/results/pubs/rr/reports/rr18_508.pdf
- vom Saal FS, Hughes C. An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment. Environ. Health Perspect. 2005. 113:926–33.
- Laura N. Vandenberg. Non-Monotonic Dose Responses in Studies of Endocrine Disrupting Chemicals: Bisphenol A as a Case Study. Dose Response. 2014 May; 12(2): 259–276.
- Liang Q, Gao X, Chen Y, Hong K, Wang HS. Cellular mechanism of the nonmonotonic dose response of bisphenol A in rat cardiac myocytes. Environ Health Perspect. 2014 Jun;122(6):601.
- Jenkins S, Wang J, Eltoum I, et al. Chronic Oral Exposure to Bisphenol A Results in a Nonmonotonic Dose Response in Mammary Carcinogenesis and Metastasis in MMTV-erbB2 Mice. Environ Health Perspect. 2011 Nov; 119(11): 1604–1609.
